Pulmotree Kolibri Mesh-Nebulizer
Improved therapy through technical progress


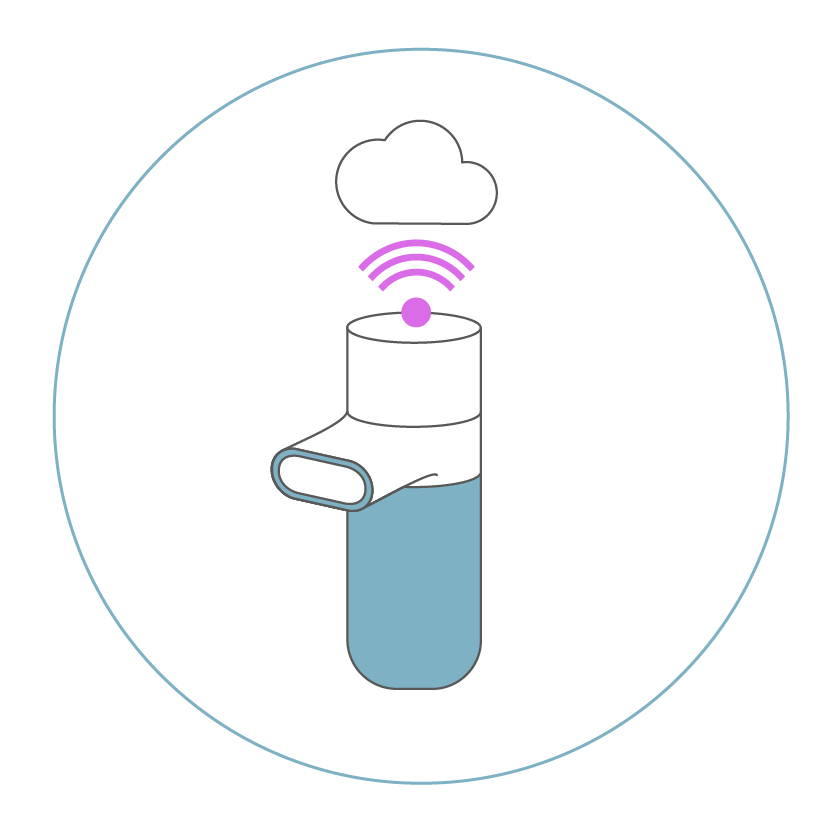
to users, physicians and CROs with the Pulmotree Connect.
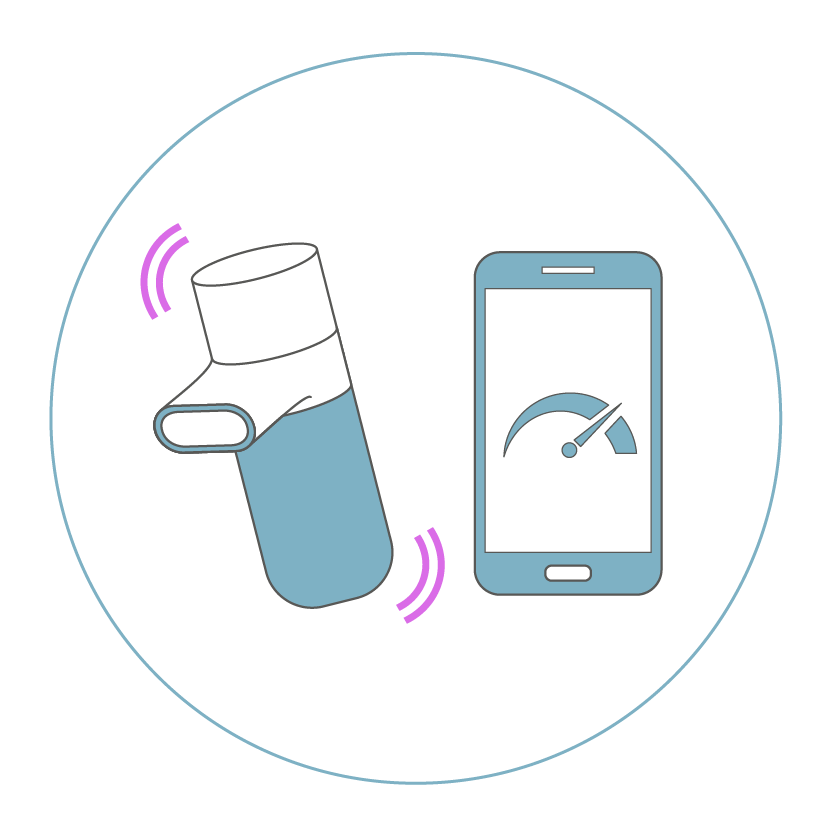
for optimized lung deposition.
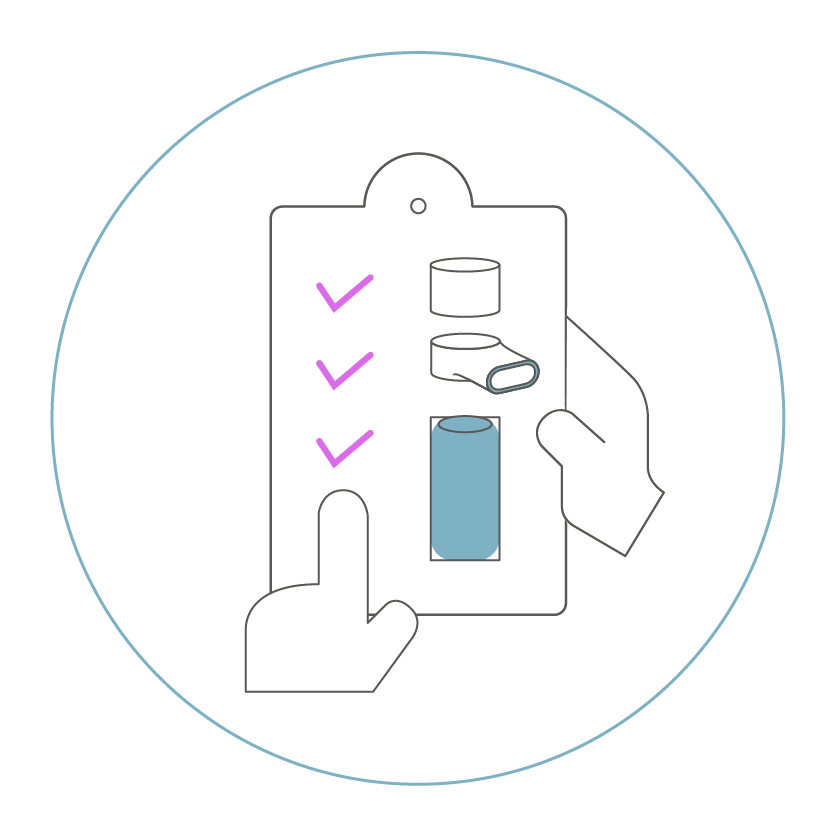
for medication, patient group and market strategy.
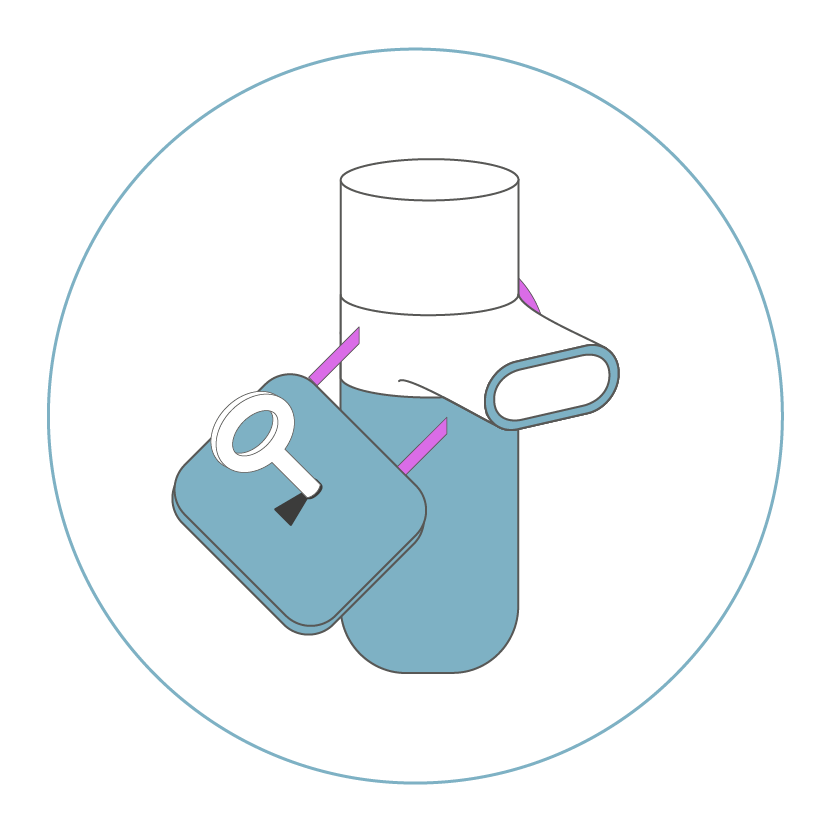
for product protection and easy handling.

for breath-triggered aerosol generation and targeted drug deposition.
The Kolibri embodies the next stage in the evolution of vibrating mesh-nebulizers, which allows an automatic and controlled support for the therapy. It provides real-time feedback on the inhalation technique and user adherence to the Pulmotree Connect app on the smartphone.
Inhalation effectiveness is increased in the long term by tracking therapy progress and providing easy-to-understand instructions for self-monitoring at home.
The Key-& Lock-System and the Closed System promote error-free inhalation, which is adapted to the user’s specific needs.


The Intelligent Administration System (INAS) helps the patient to achieve the therapeutically required inhalation flow rate and allows a targeted aerosol delivery into the intended airways.
The physician and study centers also receive separate insight into the course of therapy and can use this information to optimize the patient’s therapy plan and interpret study results more easily. In this way, the Kolibri supports improved inhalation and increases the success of the therapy with the support of experts.
The innovative design-approach of our nebulizers has enabled us to significantly reduce the amount of plastic consumables. We rely on local structures in the development and production of our Kolibri. This is particularly sustainable and also accelerates the development time.

Our goal is to minimize the factors influencing the results of clinical studies. We achieve this by being able to automatically select relevant parameters, such as the flow rate of inhalation and the patient’s inspiration time, in addition to adherence monitoring. This enables a qualitative evaluation of each individual inhalation made by the study participant, which in turn improves the interpretability of the study results. Only by understanding whether the patient has inhaled correctly can we exclude unfavorable results due to suboptimal drug deposition. And in the end, we provide an ideal low dosage of active ingredients.
We work at eye level with our partners to get the best out of each combination product. This requires first testing the core of our mesh-nebulizer for the drug and the indication and before making optimized adaptions. Once this is done, we can concentrate on equipping the nebulizer system and configuring it for the future product with an optimal cost-benefit ratio. We operate as a one-stop-shop for the therapy of respiratory diseases and support our partners in creating added value for the patient in addition to the usual development and approval activities.

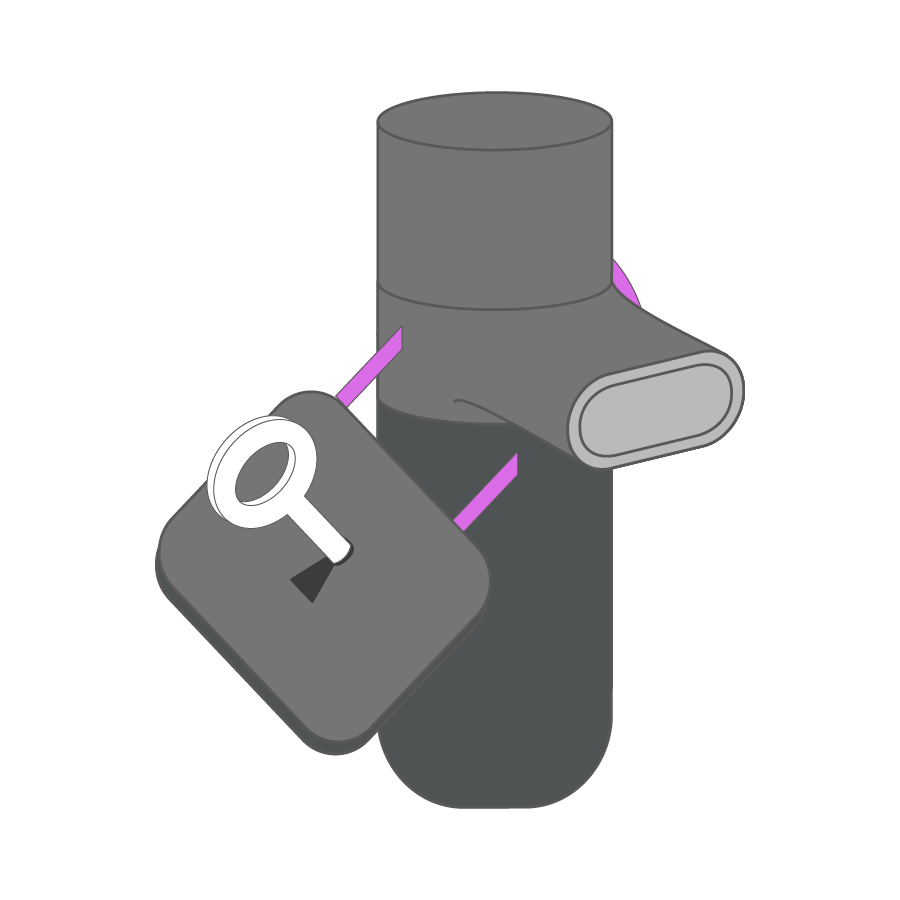
We offer solutions for patient use at home that prevent the system components from being mixed up with other Kolibri nebulizers on the market. We hereby protect our pharmaceutical partners from losses due to generic drugs, imitations or product piracy. The patient is protected against possible overdosage and can always use a functional nebulizer that is adapted to the respective medication.
Sound interesting? Then maybe we should go a step further and talk about our Closed System Concept, whereby the nebulizer system can only be used with the approved drug.
What if we could start the development of mesh-nebulizers all over again from the very beginning. And we could do so without having to sacrifice the expertise we have built up in this technology.
We have changed our perspective and asked ourselves what really helps the patient in their therapy and how we can both support the doctor in their treatment and create special added value for our pharmaceutical partners. And we have implemented this together with them.
Isn’t it fantastic when all sides really benefit from it?

